Simulating the conditions in the uterus. IVF laboratory controls
Specific management and quality control systems are key factors in increasing success rates at in vitro fertilisation laboratories (IVF laboratories).
Since 2006, all Instituto Bernabeu clinics have adhered to official international ISO 9001 quality management system standards. Furthermore, since 2015, they have implemented the specific UNE 179007-2013 Spanish standard for quality management at assisted reproduction laboratories.
These certifications guarantee that andrology, embryology and cryopreservation laboratories meet levels of excellence in:
- facilities and teams of technicians
- management
- transparency
- process traceability
Appropriate management of these systems ensures thorough controls and safety across all activities.
All our IVF laboratories must have stable environmental conditions that replicate physiological conditions as closely as possible to ensure viability of oocytes, spermatozoa and embryos. It is the reason why laboratory staff work in an environment with certain very concrete characteristics such as:
- decreased intensity of lights
- stable room temperature and humidity levels
- Positive pressure, which prevents the entry of other rooms air and particles as well as the use of absolute filters to ensure air quality and reduce levels of volatile organic compounds (VOCs), which could compromise the embryos’ development.
- Heated surfaces
State-of-the-art time-lapse incubators
In order to improve embryo development, all Instituto Bernabeu sites have incorporated new time-lapse incubators in which the embryos of each patient are individually monitored 24 hours a day. This allows embryologists to assess their development without having to remove them from the incubator, avoiding manipulation and exposure to light and maintaining stable culture conditions. Moreover, in case of any deviation in any of the critical parameters, such as temperature and/or gas levels like CO2 and O2, these incubators have alarm systems, which inform us and allow immediate correction, avoiding a possible negative impact on the development of the embryos.
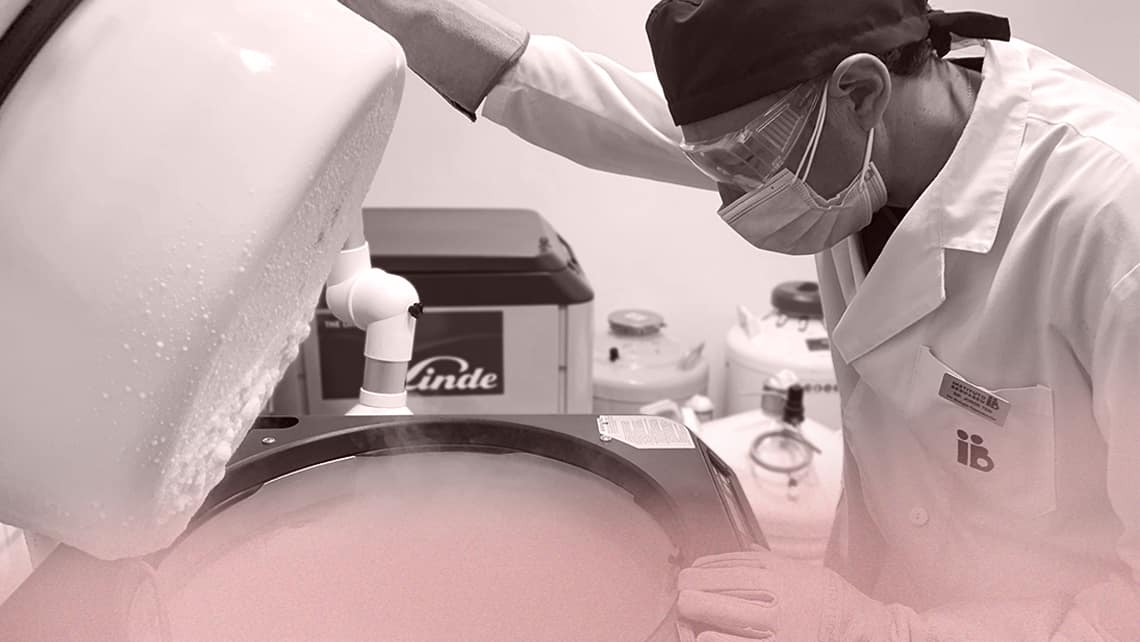
Controls at the cryogenic bank
Cryopreserved gametes and embryos are stored clearly identified in cryogenic containers with liquid nitrogen at -196°C. During the storage and handling of cryopreserved material, appropriate and safe conditions must be maintained, which means that temperatures should never exceed the critical threshold of -130°C.
Cryogenic tanks are monitored by means of manual and automatic detection and the nitrogen level and temperature recording systems of each cryobiological container. They are equipped with alarm systems that allow early detection of any situation where temperature or liquid nitrogen levels could reach values close to the safety limits, and to correct it in time. They are equipped with alarm systems to detect any situation in which the temperature or levels of liquid nitrogen are out of range of safe conditions.
For better control, the nitrogen tanks of our Bank are equipped with self-filling systems to guarantee the maintenance of cryogenic temperatures. This system is based on the use of probes that control the level of nitrogen and the temperature, which in the event of detecting a deviation send a signal to a central unit that activates the automatic filling of the tanks.
Emergency plan
It’s also important to draw up contingency plans for different emergency situations that may arise.
- In the event of power failure, there’re uninterruptible power supply (UPS) systems to back it up. This system would provide sufficient autonomy until supply is restored.
- In the event of failure of the automatic filling system, warning systems would rise and the tanks could be filled manually.
- A reserve tank of nitrogen is always available in case of any supply limitation.
Dr. Jorge Ten, director of the Embryology Unit of Instituto Bernabeu
Jaime Guerrero, Egg Donation Operational Unit Director in Instituto Bernabeu.
