Preimplantation genetic diagnosis without biopsy non-invasive: niPGT-A
Índice
What is PGD without biopsy (niPGT-A)?
The goal of in vitro fertilization is to have a healthy child. For this purpose, in the last decades different methods have been developed to perform a genetic diagnosis of the embryos (PGD) before transferring them to the uterus. These methods have been implemented to differentiate chromosomally normal embryos that give rise to on-going pregnancies and, as a consequence, healthy children from abnormal embryos (aneuploids) that do not implant, end in miscarriages or give rise to offspring with malformations.
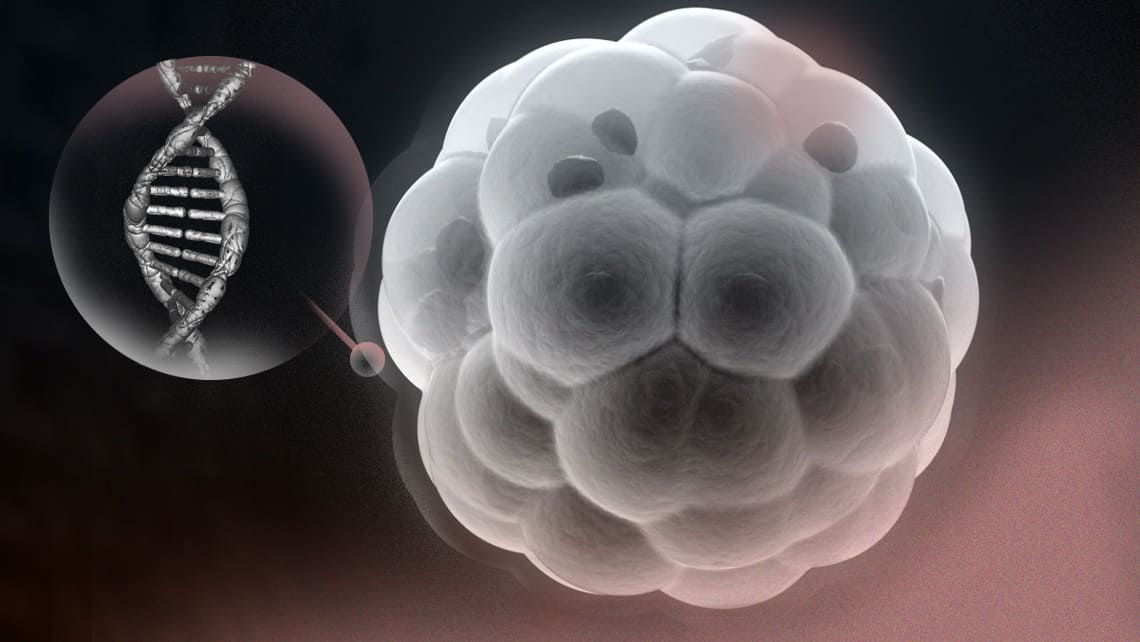
Methods used for this purpose have evolved from taking cells from the oocyte or the embryo on day 3 of its development to biopsying cells forming the outer layer of the embryo (so called, trophoectoderm) on day 5, 6 or 7 of the development. Although these techniques are safe, they require the extraction of cells (so called, “embryo biopsy”), therefore are considered invasive.
Recently it has been discovered that the growth medium where the embryo is kept, is rich in embryo’s self DNA. Therefore, research is being carried out on the possibility of making a chromosomal diagnosis of the embryo by analysing the DNA present in this culture medium. This novel technique of pre-implantation diagnosis is known as niPGT-A (Non-invasive preimplantation genetic aneuploidy test).
What does Non-invasive PGT-A consist of?
The niPGT-A or non-invasive preimplantation genetic test consists of keeping the embryo in the growth medium until day 5 or 6 of its development (until its blastocyst stage) and collecting the medium in order to carry out the analysis and draw the diagnosis. The DNA extracted from the culture medium is amplified and analyzed by mass sequencing (NGS). Simultaneously, the embryo is vitrified until the result of the analysis is obtained. The objective is to differentiate embryos that have two copies of each chromosome (normal embryos) from those that have additional ones (trisomies) or are incomplete (monosomies) on one or more chromosomes. Abnormal embryos would be discarded and normal embryos transferred to the mother’s womb.
Advantages of non-invasive genetic diagnosis (niPGT-A)
The great advantage of this new preimplantation diagnostic technique over those used until now is that it is not invasive, that is, it is not necessary to extract cells from the embryo to carry it out. Therefore, any damage that the embryo might suffer during the biopsy is avoided.

Current status of non-invesive preimplantation genetic diagnosis without biopsy (niPGT -A)
This new technique is not used in all the cases as it is being improved in order to reach 100% agreement with the results obtained via, already used, embryo biopsy confirming, this way, that the result is completely reliable. However, it will undoubtedly form part of the close future embryo genetic diagnosis and will become a new milestone in this field.
As a leading centre in genetic techniques for pre-implantation diagnosis, Instituto Bernabeu has focused a large part of its research on developing and perfecting this new method which will be the future of embryo diagnosis. It has different R+D+I projects in this area with publications in international medical journals (Reproductive BioMedicine Online) and communications at national and international conferences on Human Reproduction (ESHRE 36th Annual Meeting, 2020; 11th ASEBIR Congress in Toledo 2021).
Bibliography from Instituto Bernabeu
Consistent results of non-invasive PGT-A of human embryos using two different techniques for chromosomal analysis. Lledo B, Morales R, Ortiz JA, Rodriguez-Arnedo A, Ten J, Castillo JC, Bernabeu A, Aparicio. Ll. J, Bernabeu R. Reprod Biomed Online. 2021 Mar;42(3):555-563. doi: 10.1016/j.rbmo.2020.10.021.
Dr Ruth Morales, molecular biologist at Instituto Bernabeu.
